What Is Radio Equipment Directive (RED)?
RED Essential Requirements
The technical requirements of the European Directives appear in the form of Essential requirements, included in Article 3 of Directive 2014/53/EU. Radio equipment shall be constructed so as to ensure:
- Article 3.1(a): the protection of the health and safety of people and of domestic animals and the protection of property, including the objectives regarding safety requirements set out in Directive 2014/35/EU, but with no applicable voltage limit;
- Article 3.1(b): an adequate level of electromagnetic compatibility as set out in Directive 2014/30/EU.
- Article 3.2: that it effectively uses and supports the efficient use of radio spectrum in order to avoid harmful interference.
- Article 3.3: other requirements for certain categories or classes of Radio Equipment, including cybersecurity ones.
- (d) radio equipment does not harm the network or its functioning nor misuse network resources, thereby causing an unacceptable degradation of service.
- (e) radio equipment incorporates safeguards to ensure that the personal data and privacy of the user and of the subscriber are protected.
- (f) radio equipment supports certain features ensuring protection from fraud.
- (g) radio equipment supports certain features ensuring access to emergency services.
- Article 3.4 Common Charger (Commission Delegated Regulation (EU) 2023/1717)
How To Comply with the Radio Equipment Directive
There are three ways in which manufacturers and importers can ensure that a product or product range complies with the key requirements of the Directive or the essential requirements. Two of these require the intervention of a Notified Body.
- Module A – Internal production control: The manufacturer is responsible for drawing up the technical documentation (which must include proof of conformity testing to the relevant product standards), carrying out an internal production control and issuing a declaration of conformity. This module is only available where products have been tested, completely, to Harmonised Standards.
- Modules B + C – EU-type examination (B) and Conformity to type based on internal production control (C): These modules make the same demands on the manufacturer as module A, but a Notified Body is required (for module B) to examine the resulting technical documentation and to issue an EU-type examination certificate for the product in question. The use of these modules is mandatory for the introduction into the EU market of any product that has not been tested, completely, to Harmonised Standards. They can also be used, on a voluntary basis, for products that have been tested to Harmonised Standards. Either way, they may only be used for the conformity assessment of a product type.
- Module H – Full quality assurance: The manufacturer implements and manages a quality system encompassing radio equipment design, testing and manufacturing processes, which is subsequently assessed, certified and monitored by a Notified Body through periodic audits of the manufacturing plant. This module permits the conformity testing of a whole range of products, even of several different product ranges, and also allows the Notified Body number to be shown alongside the product’s CE Marking.
Many countries worldwide that do not have their own Radio Type Approval regulations refer to European regulations (2014/53/EU), and require an EU-type examination certificate. Therefore when addressing a worldwide homologation project, the Modules B + C approach is recommended.
The European Certification validity is permanent unless there is a change in the regulations that affects to the specific directive that have been evaluated.
Labelling Requirements
Once a product has been proved to comply with the essential requirements of the applicable Directives by means of one of the three paths defined above, the product shall be labelled with the CE mark:

EU Type Examination Certificates: a Passport to Other Markets
Many countries worldwide that do not have their own Radio Type Approval regulations refer to European regulations (2014/53/EU), and require an EU-type examination certificate. Therefore, when addressing a worldwide homologation project, the Modules B + C approach is recommended.
Notified Body Services for RED
Applus+ Laboratories is a Notified Body for the Radio Equipment Directive, (NB number 0370) for all its essential requirements:
- For Modules B+C, Applus+ Laboratories evaluates the Technical File and issues an EU Type Examination Certificate for the evaluated product.
- For Module H, the product family is registered in the Applus+ Laboratories QA records and marked by the manufacturer with the Applus+ Notified Body number.
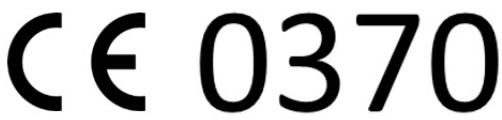
New Cybersecurity Requirements for the RED
Deadlines and Compliance Routes
We were one of the first Notified Bodies to be accredited to certify the new cybersecurity requirements or the Radio Equipment Directive, that took effect from February 1, 2022, and will be mandatory from August 1, 2025 (the EU extended the transition period 12 months in July 2023).
On January 2025, the European Commission harmonized EN 18031 standards for the Radio Equipment Directive (RED) cybersecurity articles, with restrictions. Therefore, the manufacturer can use EN18031 for self-assessment (Module A) if they:
- Fully implement EN 18031 in their product design and cybersecurity measures.
- The restrictions do not apply to their product according to the Commission Implementing Decision (EU) 2025/138 of 28 January 2025.
While self-declaration is an option (Module A) relying on expert evaluation can significantly reduce the risk of potential compliance issues down the line. If none of the routes indicated below are possible then manufacturers must undergo a conformity assessment by a Notified Body (NB).
Is My Product Affected by the New Cybersecurity Essential Requirement of the RED?
Depending on the product characteristics, manufacturers will be required to meet different essential requirements. Check this infographic or contact us to understand if your product is affected and ask for our comprehensive evidence checklist to determine whether you have all the necessary evidence in place.
As both RED Notified Body and Cybersecurity lab accredited for EN 18031, we fully support manufacturers in whatever route they decide to follow. We can provide GAP Analysis services, conduct evaluations against the EN 18031, for one or more RED articles, and provide conformity assessments as Notified Body.
For more information on the RED directive and the new Cybersecurity requirements you can read those articles on Cybersecurity Compliance for RED and EN 18031 or watch an in-depth analysis in the following video (30 min):
Why Choose Applus+ Laboratories for Radio Equipment Directive (RED) 2014/53/EU Services?
Applus+ Laboratories stands out as a trusted partner for manufacturers seeking RED compliance and certification. With extensive experience and a broad scope of accreditation, we offer:
- Full coverage of RED essential requirements including new cybersecurity provisions
- A range of conformity assessment modules including A, B+C, and H
- Expert technical evaluations and gap analysis services
- Global recognition through our EU-type examination certificates
.2025-01-10-17-04-07.png)